Ne Cation Or Anion
A negatively charged ion, as one attracted to the anode in electrolysis. Any negatively charged atom or group of atoms (opposed to cation). KOH anions and cation compound - 5420792 1s2 2s2 2p6, electron configuration for which of these ions below? There are multiple answers, choose all that applies: a Ne b Mg+2 c Na+1 d F- Make s.
- Is Ne A Cation Or Anion
- Neon Cation Or Anion
- Would You Be A Cation Or Anion
- Ne Cation Or Anion
- Be Cation Or Anion
Learning Objectives
By the end of this section, you will be able to:
- Describe and explain the observed trends in atomic size, ionization energy, and electron affinity of the elements
The elements in groups (vertical columns) of the periodic table exhibit similar chemical behavior. This similarity occurs because the members of a group have the same number and distribution of electrons in their valence shells. However, there are also other patterns in chemical properties on the periodic table. For example, as we move down a group, the metallic character of the atoms increases. Oxygen, at the top of group 16 (6A), is a colorless gas; in the middle of the group, selenium is a semiconducting solid; and, toward the bottom, polonium is a silver-grey solid that conducts electricity.
As we go across a period from left to right, we add a proton to the nucleus and an electron to the valence shell with each successive element. As we go down the elements in a group, the number of electrons in the valence shell remains constant, but the principal quantum number increases by one each time. An understanding of the electronic structure of the elements allows us to examine some of the properties that govern their chemical behavior. These properties vary periodically as the electronic structure of the elements changes. They are (1) size (radius) of atoms and ions, (2) ionization energies, and (3) electron affinities.
Variation in Covalent Radius
The quantum mechanical picture makes it difficult to establish a definite size of an atom. However, there are several practical ways to define the radius of atoms and, thus, to determine their relative sizes that give roughly similar values. We will use the covalent radius (Figure 1), which is defined as one-half the distance between the nuclei of two identical atoms when they are joined by a covalent bond (this measurement is possible because atoms within molecules still retain much of their atomic identity). We know that as we scan down a group, the principal quantum number, n, increases by one for each element. Thus, the electrons are being added to a region of space that is increasingly distant from the nucleus. Consequently, the size of the atom (and its covalent radius) must increase as we increase the distance of the outermost electrons from the nucleus. This trend is illustrated for the covalent radii of the halogens in Table 1 and Figure 1. The trends for the entire periodic table can be seen in Figure 1.
| Table 1. Covalent Radii of the Halogen Group Elements | ||
|---|---|---|
| Atom | Covalent radius (pm) | Nuclear charge |
| F | 64 | +9 |
| Cl | 99 | +17 |
| Br | 114 | +35 |
| I | 133 | +53 |
| At | 148 | +85 |
Figure 1. (a) The radius of an atom is defined as one-half the distance between the nuclei in a molecule consisting of two identical atoms joined by a covalent bond. The atomic radius for the halogens increases down the group as n increases. (b) Covalent radii of the elements are shown to scale. The general trend is that radii increase down a group and decrease across a period.
Figure 2. Within each period, the trend in atomic radius decreases as Z increases; for example, from K to Kr. Within each group (e.g., the alkali metals shown in purple), the trend is that atomic radius increases as Z increases.
As shown in Figure 2, as we move across a period from left to right, we generally find that each element has a smaller covalent radius than the element preceding it. This might seem counterintuitive because it implies that atoms with more electrons have a smaller atomic radius. This can be explained with the concept of effective nuclear charge, Zeff. This is the pull exerted on a specific electron by the nucleus, taking into account any electron–electron repulsions. For hydrogen, there is only one electron and so the nuclear charge (Z) and the effective nuclear charge (Zeff) are equal. For all other atoms, the inner electrons partially shield the outer electrons from the pull of the nucleus, and thus:
[latex]{Z}_{text{eff}}=Z-text{shielding}[/latex]
Shielding is determined by the probability of another electron being between the electron of interest and the nucleus, as well as by the electron–electron repulsions the electron of interest encounters. Core electrons are adept at shielding, while electrons in the same valence shell do not block the nuclear attraction experienced by each other as efficiently. Thus, each time we move from one element to the next across a period, Z increases by one, but the shielding increases only slightly. Thus, Zeff increases as we move from left to right across a period. The stronger pull (higher effective nuclear charge) experienced by electrons on the right side of the periodic table draws them closer to the nucleus, making the covalent radii smaller.
Thus, as we would expect, the outermost or valence electrons are easiest to remove because they have the highest energies, are shielded more, and are farthest from the nucleus. As a general rule, when the representative elements form cations, they do so by the loss of the ns or np electrons that were added last in the Aufbau process. The transition elements, on the other hand, lose the ns electrons before they begin to lose the (n – 1)d electrons, even though the ns electrons are added first, according to the Aufbau principle.
Example 1: Sorting Atomic Radii
Predict the order of increasing covalent radius for Ge, Fl, Br, Kr.
Show AnswerRadius increases as we move down a group, so Ge < Fl (Note: Fl is the symbol for flerovium, element 114, NOT fluorine). Radius decreases as we move across a period, so Kr < Br < Ge. Putting the trends together, we obtain Kr < Br < Ge < Fl.
Check Your Learning
Give an example of an atom whose size is smaller than fluorine.
Variation in Ionic Radii
Ionic radius is the measure used to describe the size of an ion. A cation always has fewer electrons and the same number of protons as the parent atom; it is smaller than the atom from which it is derived (Figure 3). For example, the covalent radius of an aluminum atom (1s22s22p63s23p1) is 118 pm, whereas the ionic radius of an Al3+ (1s22s22p6) is 68 pm. As electrons are removed from the outer valence shell, the remaining core electrons occupying smaller shells experience a greater effective nuclear charge Zeff (as discussed) and are drawn even closer to the nucleus.
Figure 3. The radius for a cation is smaller than the parent atom (Al), due to the lost electrons; the radius for an anion is larger than the parent (S), due to the gained electrons.
Cations with larger charges are smaller than cations with smaller charges (e.g., V2+ has an ionic radius of 79 pm, while that of V3+ is 64 pm). Proceeding down the groups of the periodic table, we find that cations of successive elements with the same charge generally have larger radii, corresponding to an increase in the principal quantum number, n.
An anion (negative ion) is formed by the addition of one or more electrons to the valence shell of an atom. This results in a greater repulsion among the electrons and a decrease in Zeff per electron. Both effects (the increased number of electrons and the decreased Zeff) cause the radius of an anion to be larger than that of the parent atom (Figure 3). For example, a sulfur atom ([Ne]3s23p4) has a covalent radius of 104 pm, whereas the ionic radius of the sulfide anion ([Ne]3s23p6) is 170 pm. For consecutive elements proceeding down any group, anions have larger principal quantum numbers and, thus, larger radii.
Atoms and ions that have the same electron configuration are said to be isoelectronic. Examples of isoelectronic species are N3–, O2–, F–, Ne, Na+, Mg2+, and Al3+ (1s22s22p6). Another isoelectronic series is P3–, S2–, Cl–, Ar, K+, Ca2+, and Sc3+ ([Ne]3s23p6). For atoms or ions that are isoelectronic, the number of protons determines the size. The greater the nuclear charge, the smaller the radius in a series of isoelectronic ions and atoms.
Variation in Ionization Energies
The amount of energy required to remove the most loosely bound electron from a gaseous atom in its ground state is called its first ionization energy (IE1). The first ionization energy for an element, X, is the energy required to form a cation with +1 charge:
[latex]text{X}left(gright)longrightarrow {text{X}}^{text{+}}left(gright)+{text{e}}^{-}{text{IE}}_{1}[/latex]
The energy required to remove the second most loosely bound electron is called the second ionization energy (IE2).
[latex]{text{X}}^{text{+}}left(gright)longrightarrow {text{X}}^{2+}left(gright)+{text{e}}^{-}{text{IE}}_{2}[/latex]
The energy required to remove the third electron is the third ionization energy, and so on. Energy is always required to remove electrons from atoms or ions, so ionization processes are endothermic and IE values are always positive. For larger atoms, the most loosely bound electron is located farther from the nucleus and so is easier to remove. Thus, as size (atomic radius) increases, the ionization energy should decrease. Relating this logic to what we have just learned about radii, we would expect first ionization energies to decrease down a group and to increase across a period.
Figure 4 graphs the relationship between the first ionization energy and the atomic number of several elements. The values of first ionization energy for the elements are given in Figure 5. Within a period, the IE1 generally increases with increasing Z. Down a group, the IE1 value generally decreases with increasing Z. There are some systematic deviations from this trend, however. Note that the ionization energy of boron (atomic number 5) is less than that of beryllium (atomic number 4) even though the nuclear charge of boron is greater by one proton. This can be explained because the energy of the subshells increases as l increases, due to penetration and shielding (as discussed previously in this chapter). Within any one shell, the s electrons are lower in energy than the p electrons. This means that an s electron is harder to remove from an atom than a p electron in the same shell. The electron removed during the ionization of beryllium ([He]2s2) is an s electron, whereas the electron removed during the ionization of boron ([He]2s22p1) is a p electron; this results in a lower first ionization energy for boron, even though its nuclear charge is greater by one proton. Thus, we see a small deviation from the predicted trend occurring each time a new subshell begins.
Figure 4. The first ionization energy of the elements in the first five periods are plotted against their atomic number.
Figure 5. This version of the periodic table shows the first ionization energy of (IE1), in kJ/mol, of selected elements.
Another deviation occurs as orbitals become more than one-half filled. The first ionization energy for oxygen is slightly less than that for nitrogen, despite the trend in increasing IE1 values across a period. Looking at the orbital diagram of oxygen, we can see that removing one electron will eliminate the electron–electron repulsion caused by pairing the electrons in the 2p orbital and will result in a half-filled orbital (which is energetically favorable). Analogous changes occur in succeeding periods (note the dip for sulfur after phosphorus in Figure 5).
Removing an electron from a cation is more difficult than removing an electron from a neutral atom because of the greater electrostatic attraction to the cation. Likewise, removing an electron from a cation with a higher positive charge is more difficult than removing an electron from an ion with a lower charge. Thus, successive ionization energies for one element always increase. As seen in Table 2, there is a large increase in the ionization energies (color change) for each element. This jump corresponds to removal of the core electrons, which are harder to remove than the valence electrons. For example, Sc and Ga both have three valence electrons, so the rapid increase in ionization energy occurs after the third ionization.
| Table 2. Successive Ionization Energies for Selected Elements (kJ/mol) | |||||||
|---|---|---|---|---|---|---|---|
| Element | IE1 | IE2 | IE3 | IE4 | IE5 | IE6 | IE7 |
| K | 418.8 | 3051.8 | 4419.6 | 5876.9 | 7975.5 | 9590.6 | 11343 |
| Ca | 589.8 | 1145.4 | 4912.4 | 6490.6 | 8153.0 | 10495.7 | 12272.9 |
| Sc | 633.1 | 1235.0 | 2388.7 | 7090.6 | 8842.9 | 10679.0 | 13315.0 |
| Ga | 578.8 | 1979.4 | 2964.6 | 6180 | 8298.7 | 10873.9 | 13594.8 |
| Ge | 762.2 | 1537.5 | 3302.1 | 4410.6 | 9021.4 | N/A | N/A |
| As | 944.5 | 1793.6 | 2735.5 | 4836.8 | 6042.9 | 12311.5 | N/A |
Example 2: Ranking Ionization Energies
Predict the order of increasing energy for the following processes: IE1 for Al, IE1 for Tl, IE2 for Na, IE3 for Al.
Show Answer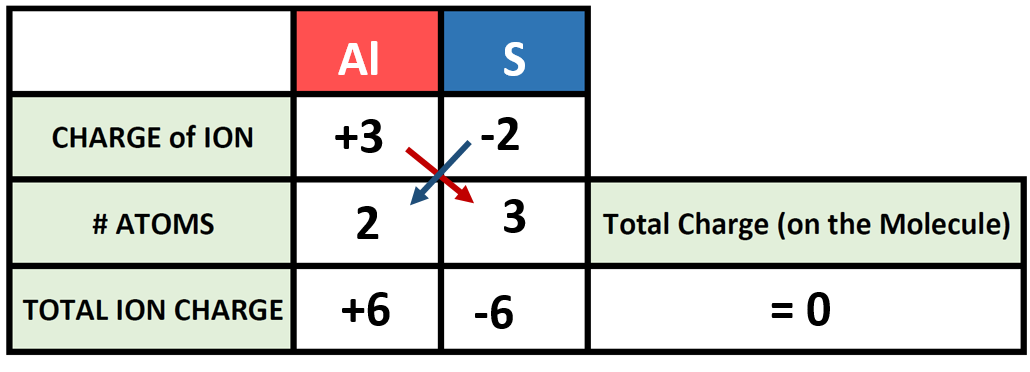

Removing the 6p1 electron from Tl is easier than removing the 3p1 electron from Al because the higher n orbital is farther from the nucleus, so IE1(Tl) < IE1(Al). Ionizing the third electron from [latex]text{Al}left({text{Al}}^{2+}longrightarrow {text{Al}}^{3+}+{text{e}}^{text{-}}right)[/latex] requires more energy because the cation Al2+ exerts a stronger pull on the electron than the neutral Al atom, so IE1(Al) < IE3(Al). The second ionization energy for sodium removes a core electron, which is a much higher energy process than removing valence electrons. Putting this all together, we obtain: IE1(Tl) < IE1(Al) < IE3(Al) < IE2(Na).
Check Your Learning
Which has the lowest value for IE1: O, Po, Pb, or Ba?
Variation in Electron Affinities
The electron affinity [EA] is the energy change for the process of adding an electron to a gaseous atom to form an anion (negative ion).
[latex]text{X}left(gright)+{text{e}}^{-}longrightarrow {text{X}}^{-}left(gright){text{EA}}_{1}[/latex]
Is Ne A Cation Or Anion
This process can be either endothermic or exothermic, depending on the element. The EA of some of the elements is given in Figure 6. You can see that many of these elements have negative values of EA, which means that energy is released when the gaseous atom accepts an electron. However, for some elements, energy is required for the atom to become negatively charged and the value of their EA is positive. Just as with ionization energy, subsequent EA values are associated with forming ions with more charge. The second EA is the energy associated with adding an electron to an anion to form a –2 ion, and so on.
As we might predict, it becomes easier to add an electron across a series of atoms as the effective nuclear charge of the atoms increases. We find, as we go from left to right across a period, EAs tend to become more negative. The exceptions found among the elements of group 2 (2A), group 15 (5A), and group 18 (8A) can be understood based on the electronic structure of these groups. The noble gases, group 18 (8A), have a completely filled shell and the incoming electron must be added to a higher n level, which is more difficult to do. Group 2 (2A) has a filled ns subshell, and so the next electron added goes into the higher energy np, so, again, the observed EA value is not as the trend would predict. Finally, group 15 (5A) has a half-filled np subshell and the next electron must be paired with an existing np electron. In all of these cases, the initial relative stability of the electron configuration disrupts the trend in EA.
We also might expect the atom at the top of each group to have the largest EA; their first ionization potentials suggest that these atoms have the largest effective nuclear charges. However, as we move down a group, we see that the second element in the group most often has the greatest EA. The reduction of the EA of the first member can be attributed to the small size of the n = 2 shell and the resulting large electron–electron repulsions. For example, chlorine, with an EA value of –348 kJ/mol, has the highest value of any element in the periodic table. The EA of fluorine is –322 kJ/mol. When we add an electron to a fluorine atom to form a fluoride anion (F–), we add an electron to the n = 2 shell. The electron is attracted to the nucleus, but there is also significant repulsion from the other electrons already present in this small valence shell. The chlorine atom has the same electron configuration in the valence shell, but because the entering electron is going into the n = 3 shell, it occupies a considerably larger region of space and the electron–electron repulsions are reduced. The entering electron does not experience as much repulsion and the chlorine atom accepts an additional electron more readily.
Figure 6. This version of the periodic table displays the electron affinity values (in in kJ/mol) for selected elements.
The properties discussed in this section (size of atoms and ions, effective nuclear charge, ionization energies, and electron affinities) are central to understanding chemical reactivity. For example, because fluorine has an energetically favorable EA and a large energy barrier to ionization (IE), it is much easier to form fluorine anions than cations. Metallic properties including conductivity and malleability (the ability to be formed into sheets) depend on having electrons that can be removed easily. Thus, metallic character increases as we move down a group and decreases across a period in the same trend observed for atomic size because it is easier to remove an electron that is farther away from the nucleus.
Key Concepts and Summary
Electron configurations allow us to understand many periodic trends. Covalent radius increases as we move down a group because the n level (orbital size) increases. Covalent radius mostly decreases as we move left to right across a period because the effective nuclear charge experienced by the electrons increases, and the electrons are pulled in tighter to the nucleus. Anionic radii are larger than the parent atom, while cationic radii are smaller, because the number of valence electrons has changed while the nuclear charge has remained constant. Ionization energy (the energy associated with forming a cation) decreases down a group and mostly increases across a period because it is easier to remove an electron from a larger, higher energy orbital. Electron affinity (the energy associated with forming an anion) is more favorable (exothermic) when electrons are placed into lower energy orbitals, closer to the nucleus. Therefore, electron affinity becomes increasingly negative as we move left to right across the periodic table and decreases as we move down a group. For both IE and electron affinity data, there are exceptions to the trends when dealing with completely filled or half-filled subshells.
Exercises
- Based on their positions in the periodic table, predict which has the smallest atomic radius: Mg, Sr, Si, Cl, I.
- Based on their positions in the periodic table, predict which has the largest atomic radius: Li, Rb, N, F, I.
- Based on their positions in the periodic table, predict which has the largest first ionization energy: Mg, Ba, B, O, Te.
- Based on their positions in the periodic table, predict which has the smallest first ionization energy: Li, Cs, N, F, I.
- Based on their positions in the periodic table, rank the following atoms in order of increasing first ionization energy: F, Li, N, Rb
- Based on their positions in the periodic table, rank the following atoms or compounds in order of increasing first ionization energy: Mg, O, S, Si
- Atoms of which group in the periodic table have a valence shell electron configuration of ns2np3?
- Atoms of which group in the periodic table have a valence shell electron configuration of ns2?
- Based on their positions in the periodic table, list the following atoms in order of increasing radius: Mg, Ca, Rb, Cs.
- Based on their positions in the periodic table, list the following atoms in order of increasing radius: Sr, Ca, Si, Cl.
- Based on their positions in the periodic table, list the following ions in order of increasing radius: K+, Ca2+, Al3+, Si4+.
- List the following ions in order of increasing radius: Li+, Mg2+, Br–, Te2–.
- Which atom and/or ion is (are) isoelectronic with Br+: Se2+, Se, As–, Kr, Ga3+, Cl–?
- Which of the following atoms and ions is (are) isoelectronic with S2+: Si4+, Cl3+, Ar, As3+, Si, Al3+?
- Compare both the numbers of protons and electrons present in each to rank the following ions in order of increasing radius: As3–, Br–, K+, Mg2+.
- Of the five elements Al, Cl, I, Na, Rb, which has the most exothermic reaction? (E represents an atom.) What name is given to the energy for the reaction? Hint: note the process depicted does not correspond to electron affinity [latex]{text{E}}^{text{+}}left(gright)+{text{e}}^{-}longrightarrow text{E}left(gright)[/latex]
- Of the five elements Sn, Si, Sb, O, Te, which has the most endothermic reaction? (E represents an atom.) What name is given to the energy for the reaction? [latex]text{E}left(gright)longrightarrow {text{E}}^{text{+}}left(gright)+{text{e}}^{-}[/latex]
- The ionic radii of the ions S2–, Cl–, and K+ are 184, 181, 138 pm respectively. Explain why these ions have different sizes even though they contain the same number of electrons.
- Which main group atom would be expected to have the lowest second ionization energy?
- Explain why Al is a member of group 13 rather than group 3?
1. Cl
3. O
5. Rb < Li < N < F
7. 15 (5A)
9. Mg < Ca < Rb < Cs
11. Si4+ < Al3+ < Ca2+ < K+
13. Se, As−
15. Mg2+ < K+ < Br– < As3–
Neon Cation Or Anion
17. O, IE1
19. Ra
Glossary
covalent radius: one-half the distance between the nuclei of two identical atoms when they are joined by a covalent bond
effective nuclear charge: charge that leads to the Coulomb force exerted by the nucleus on an electron, calculated as the nuclear charge minus shielding
electron affinity: energy required to add an electron to a gaseous atom to form an anion
ionization energy: energy required to remove an electron from a gaseous atom or ion. The associated number (e.g., second ionization energy) corresponds to the charge of the ion produced (X2+)
isoelectronic: group of ions or atoms that have identical electron configurations
Main Difference
The main difference between Atom and Ion is that Atom is the fundamental unit of matter. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas Ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge.
Atom vs. Ion
An atom is a fundamental unit of all matters in this universe, thus deriving all components. In contrast, Ion is charge bearing atom, i.e. that contains a positive or a negative charge. Atoms have no further classifications and is a fundamental unit they compose of matter founded in the universe, but, Ions further classified into two types as Cations and Anions based on their carrying a positive or negative charge.
Would You Be A Cation Or Anion
Ne Cation Or Anion
Atoms are composed of equal protons and electrons as in their existing natural forms as a whole with varying numbers of electrons and protons with varying atomic numbers. Whereas, Ions contain an odd or unequal number of electrons and protons as atom attracts an electron in their outermost shell or lose an electron to another atom to become an ion.
Atoms bear a net neutral charge due to the equal number of protons that carry a positive charge and electrons that carry a negative charge. While Ions bear a net positive or a negative charge due to the unequal number of electrons and protons, i.e. they attract an electron in their outermost shell or lose an electron to another atom. Atoms do not bear any electrical charge, i.e. net neutral charge thus does not show any attraction or repulsion to electric fields. However, as Ions carry a net positive or a negative charge; therefore, they are attractive or repulsive to electric fields as per their bearing charge.
Atoms denoted by a simple molecular formula of an element are represented neutral due to no charge. In contrast, Ions are denoted by a charge either negative or positive, representing the number of excess or lost electrons as per their carrying charge. Atoms have examples as Neon is represented by Ne and Iron is represented by Fe and respectively, whereas, Ions have examples as Cobalt ion is denoted by Co 2+ and Nitride – N3- and respectively.
Comparison Chart
| Atom | Ion |
| The fundamental unit of matter in this universe contains an equal number of electrons and protons. | Atoms that either contains a negative or a positive charge. |
| Classifications | |
| No further Classification | Classified into Cations and Anions |
| Number of Electrons | |
| Equal to the number of Protons | Not Equal to the number of Protons |
| Electrical Charge | |
| Neutral | Positive or Negatively charged |
| Reaction to Electric Field | |
| No reaction | Attracted or repulsed |
| Presentation | |
| Neutral Formula of Atom | The formula of Atom with respect to bearing charge |
| Examples | |
| Neon as Ne, Iron by Fe, etc. | Cobalt ion as Co 2+ and Nitride as N3- |
What is an Atom?
Atoms are the fundamental unit of all matters in this universe as they derive all components. In early times, scientists believed that Atoms could not further divide, which was voided by the discovery of protons, electron, and nucleus. The atomic theory describes the structure of an Atom as how an Atom is composed of electrons and protons. In modern times, this theory is preceded by modern atomic theory according to which Atoms composed of two components. These components are the nucleus and subatomic particles.
Atoms have no further classifications and being a fundamental unit they compose of matter founded in the universe. And as described above, composed of equal protons and electrons as in their existing natural forms with varying atomic number. Atoms bear as a result net neutral charge due to the equivalent number of protons that carry a positive charge and electrons that carry a negative charge. They also do not show any attraction or repulsion to electric fields.
Be Cation Or Anion
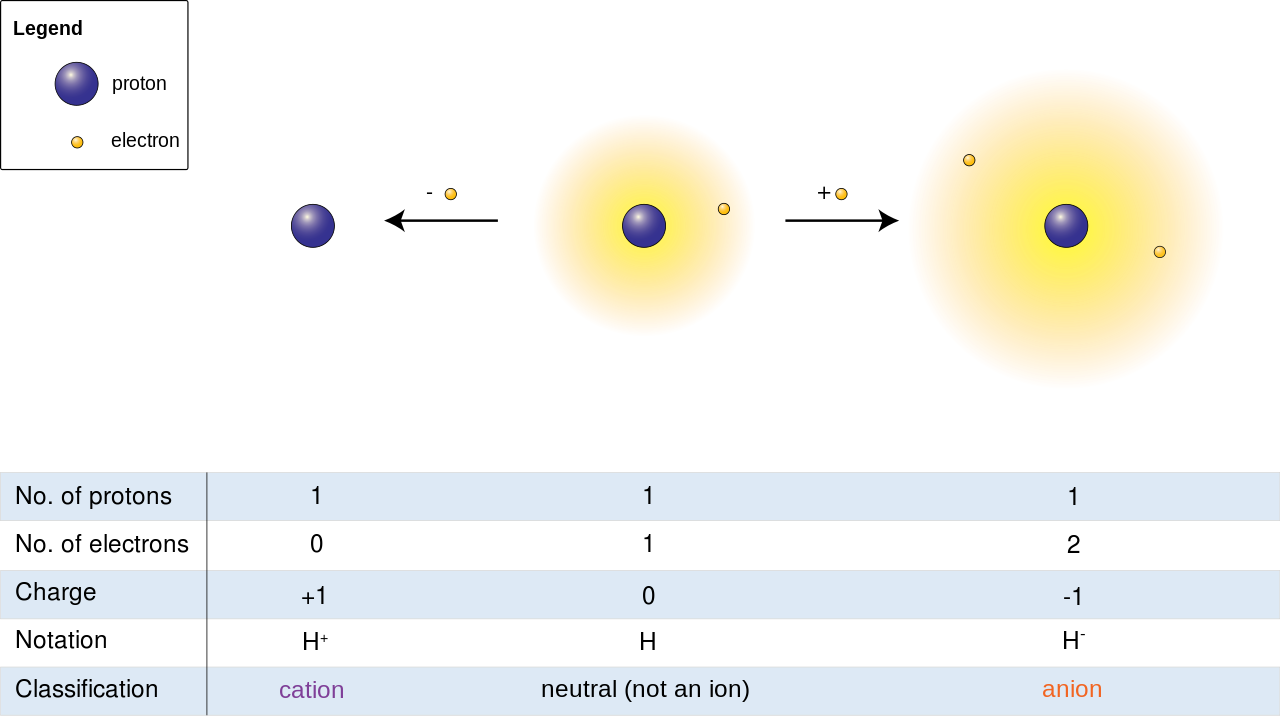
Atoms can form chemical bonds with each other to form molecules and compounds. These bonds can either be a covalent bond, ionic bond, or metallic bonds. These bonds are formed between the Atoms due to the exchange of electrons with each other.
However, protons and neutrons do not directly take part in bond formation. Atoms denoted by a simple molecular formula of an element and are represented neutral due to no charge. Some common examples of Atoms include Neon Atoms, which are represented by Ne and Hydrogen, which is represented by H and, respectively.
What is Ion?
Ion is an Atom or Molecules that bear a net charge, i.e. that contain a positive or a negative charge. Because when an Atom or a Molecule attracts an electron in their outermost shell or loses an electron to another Atom, it becomes an Ion. They further classified into two types as Cations and Anions based on their carrying a positive or negative charge.
Anions are the Ions that bear a net negative charge by gaining excess electrons from their surrounding atoms or molecules because the protons in the nucleus get unable to neutralize the impact of gained electrons. Cations are the kind of atoms or molecules that, by losing electrons from their outermost shell exposed to the environment, bear a net positive charge.
Ions as bear a net positive or a negative charge, thus show attraction or repulsion towards electric fields as per their bearing charge. They are also able to form ionic bonds. These ionic bonds are formed in response to these attracted ions, which are attracted to each other by the electrostatic force of attraction because of two opposite charges on these atoms. These Ions are denoted by a charge, either negative or positive, representing the number of excess or lost electrons. Common examples of Ions are, Cobalt that is denoted by Co 2+, and Nitride that is denoted by N3- and respectively.
Key Differences

- An atom is a fundamental unit of all matters in this universe; on the contrary, Ions are positive or negative charge bearing atoms.
- Atoms are composed of equal protons and electrons as in their existing natural forms; on the flip side, Ions contain an unequal number of electrons and protons by attracting or losing an electron in their outermost shell.
- Atoms bear a net neutral charge due to an equal number of protons and electrons, on the other hand, Ions bear a net positive or a negative charge due to an unequal number of electrons and protons.
- Atoms do not show any attraction or repulsion to an electric field, whereas, as Ions bear a net positive or a negative charge, they show attraction or repulsion to electric fields as per their bearing charge.
- Atoms are denoted by a neutral molecular formula of an element due to no charge. On the contrary, Ions are denoted by a charge either negative or positive as per the number of electrons with respect to protons.
- Atoms have no further classifications, on the flip side, Ions further classified into two types as Cations and Anions based on their carrying a positive or negative charge.
- Atoms have examples as the Hydrogen atom represented by H and Iron is represented by Fe, whereas the example of Ions is; Cobalt, which is denoted by Co 2+ and Nitride, which is denoted by N3-.
Conclusion
Atoms are the fundamental unit of matter that contains an equal number of electrons and protons and bears a net neutral electrical charge. However, Ions composed of atoms that bear a net positive or negative charge due to an unequal number of protons and electrons.